Pipeline
Clene is advancing a pipeline of nanotherapeutics to address high unmet medical need in several disease areas.
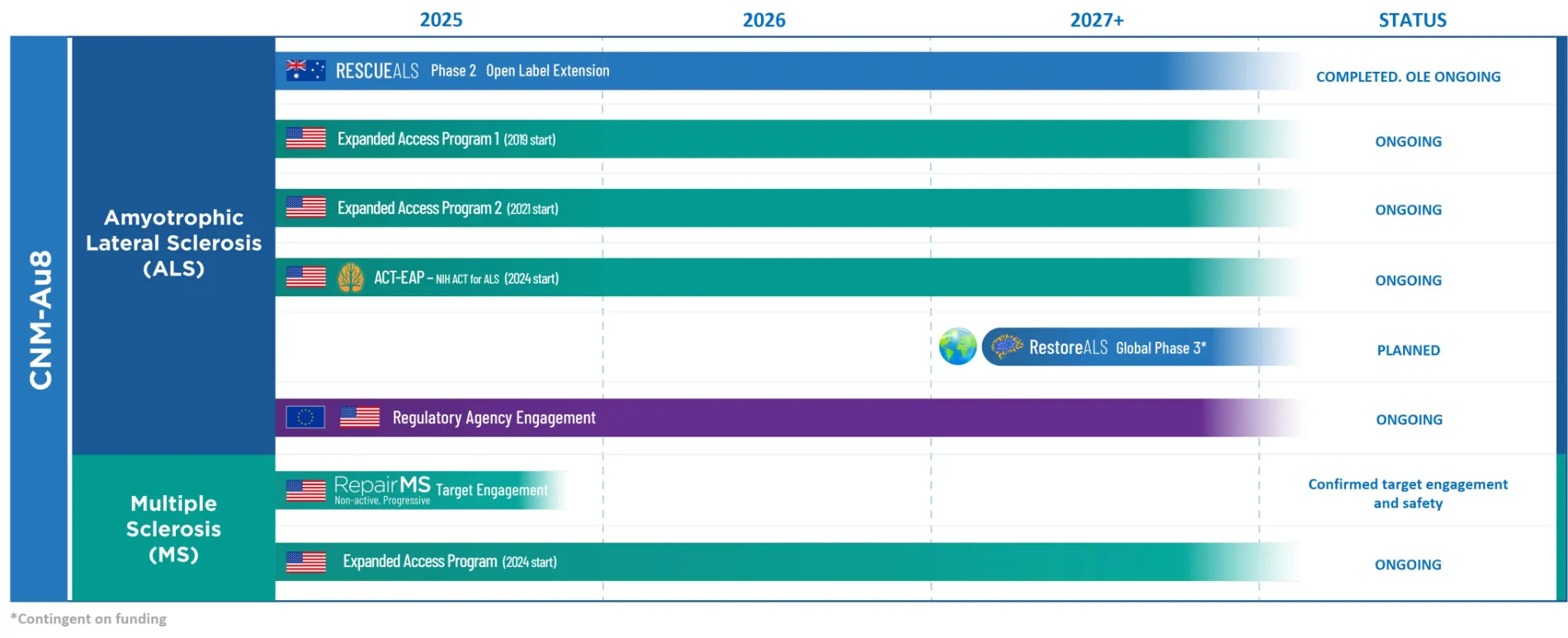
CNM-Au8®
CNM-Au8® is a gold nanocrystal suspension currently in development as a disease-modifying treatment for people living with Amyotrophic Lateral Sclerosis (ALS), Multiple Sclerosis (MS), and Parkinson’s Disease (PD).
For additional information on Clene’s trials of CNM-Au8® in ALS:
- HEALEY-ALS Platform: NCT04414345
- RESCUE-ALS: NCT04098406
- EAP: NCT04081714
- EAP: NCT05281484
For additional information on Clene’s trials of CNM-Au8® in MS:
- REPAIR-MS: NCT03993171
- VISIONARY-MS: NCT03536559
- VISIONMS-LTE: NCT04626921
For additional information on Clene’s trial of CNM-Au8® in PD:
- REPAIR-PD: NCT03815916
CNM-ZnAg
CNM-AgZn17
CNM-PtAu7
About Clinical Trials
A clinical trial is a research study used to validate a new treatment or therapy in volunteers. Through clinical trials, researchers and healthcare professionals are able to determine if a treatment is safe and effective, and how it can be used to treat a specific disease or condition.
By participating in a clinical trial, you are taking an active role in the development and approval of new medicines and treatments. People who volunteer in clinical trials provide the medical community with valuable research and data that helps to facilitate the development of new and potentially life-saving therapies.











